
September 27, 1999
UCSC researchers obtain first detailed images of a complete ribosome
By Tim Stephens
Researchers in the Center for the Molecular Biology of RNA have taken a huge step
toward solving one of the most fundamental and daunting problems of molecular biology
by obtaining images of the complete structure of the ribosome, a key component of
all living things. Knowing the structure of the ribosome may lead to more effective
antibiotics, new insights into the origins of life, and other significant advances.
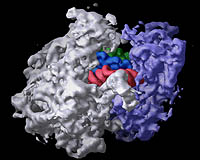 |
| The bacterial ribosome is composed of three different RNA molecules and more than
50 different proteins arranged in two major subunits, which join together to form
the complete ribosome. (More) Image: Center for Molecular Biology of RNA |
 |
| Harry Noller and members of his research team, Marat Yusupov and Gulnara Yusupova,
in their laboratory. Credit: UCSC Photography Services |
Ribosomes are tiny molecular machines responsible for translating the genetic code and synthesizing proteins. They are the link between the genetic instructions encoded in DNA and the protein molecules that carry out the activities of living cells.
The images obtained by the UCSC researchers show how different parts of the ribosome interact with one another and how the ribosome interacts with certain molecules involved in protein synthesis. Although researchers are eager to obtain even more detailed images of the ribosome in action, these results show that the ultimate goal of understanding exactly how the ribosome works is finally within reach.
"What we have at present are a few snapshots, and ultimately what we would like is a movie of the ribosome in action," said Harry Noller, Sinsheimer Professor of Molecular Biology and head of the group that obtained the new images. Their findings are reported in two papers published in the September 24 issue of the journal Science.
The potential applications of this research cover a broad range, from understanding the origins of life to developing more effective antibiotics, Noller said. Ribosomes are ancient structures that show little variation among different forms of life. Inside every living cell, tens of thousands of ribosomes churn out proteins with mind-boggling speed and precision.
Most research has focused on bacterial ribosomes, which are a bit smaller than those in higher organisms. Even bacterial ribosomes are extraordinarily complex. They are composed of three different RNA molecules and more than 50 different proteins arranged in two major subunits, which join together to form the complete ribosome.
To obtain images of this complex structure, Noller's group used a technique called x-ray crystallography. This involved growing crystals of purified ribosomes, shining a focused beam of x-rays through the crystals, and analyzing the resulting diffraction pattern. The complete ribosome is the largest molecular structure ever solved by x-ray crystallography. (Some virus structures of comparable size have also been solved, but their symmetry allowed scientists to extrapolate from results obtained from a small portion of the whole structure.)
Noller, who directs UCSC's Center for the Molecular Biology of RNA, has been studying the ribosome for the past 30 years. His coauthors include postdoctoral researchers Jamie Cate, now on the faculty of the Whitehead Institute for Biomedical Research in Cambridge, Massachusetts, and Gloria Culver, now an assistant professor at Iowa State University; visiting researchers Marat Yusupov and Gulnara Yusupova; and Thomas Earnest, director of the crystallography facility at Lawrence Berkeley National Laboratory, one of a handful of sites with a synchrotron capable of producing the high-energy x-rays needed for crystallography of a structure as large as the ribosome.
In the first and longer of the two papers, the researchers present the structure of the complete ribosome with several of the molecules involved in protein synthesis bound to it. The crystals used to obtain this structure were prepared by the husband-and-wife team of Yusupov and Yusupova, who have been working on ribosome crystallization techniques for more than ten years. The crystallographic analysis, performed by Cate, yielded a medium-resolution structure, not detailed enough to show the location of every atom but sufficient to provide valuable insights into the ribosome's mechanism of action.
Cate compared the ribosome to a miniature factory where proteins are made on an assembly line. "To a large extent it has been a mystery how this assembly line is laid out, and these papers show where different pieces of equipment are on the shop floor," he said.
Biologists have known the basic outlines of protein synthesis for decades. The instructions for making a protein are carried to the ribosome by a messenger RNA molecule, which has copied the instructions from chromosomal DNA, the storehouse of genetic information carried in every cell. The building blocks of proteins are carried to the ribosome by transfer RNA molecules. On the ribosome, the transfer RNAs recognize specific sequences of genetic code on the messenger RNA, and the protein building blocks are then joined together in the proper order.
There are three distinct binding sites for transfer RNAs on the ribosome, and the new images show how the transfer RNAs are positioned in each of those binding sites. During protein synthesis, the transfer RNAs pass sequentially through the three binding sites, which are located at the interface between the two subunits of the ribosome.
"The transfer RNAs come through as if on a conveyor belt, and we can see how the ribosome holds the transfer RNA differently in each of the binding sites," Cate said.
Another major finding is the recognition of what appears to be a molecular relay mechanism that may be part of a communication pathway between the large subunit and the small subunit. One of the elements of this pathway is a coil of RNA previously identified by other researchers as a molecular "switch" that can move between two different conformations.
"The ribosome appears to be a dynamic molecular machine with moving parts and a very complicated mechanism of action," Noller said.
The researchers presented additional details of the connections between the two subunits of the ribosome in the second paper. That paper describes a molecular bridge in which a loop of ribosomal RNA projects from the large subunit and interacts with a protein component of the smaller subunit.
"Understanding how the subunits interact is critical to understanding how the ribosome works, and for the first time we've been able to identify the protein and RNA components of a bridge between the two subunits," said Culver, who performed a series of biochemical studies to characterize the components of the bridge.
"The x-ray crystallography and the biochemical experiments complemented each other beautifully to provide these very clear results," Noller noted.
The papers from Noller's lab follow close on the heels of reports published last month in the journal Nature by two other groups who used x-ray crystallography to study the structures of each of the two major subunits of the ribosome. Cate said he was struck by how little overlap there was between the findings reported by the three groups. Whereas the Nature papers focused on the architecture of the individual subunits, the UCSC researchers showed how the two subunits are joined and sought to interpret the images in terms of how the ribosome functions.
"It shows just how wide open this field is that three groups can come up with completely different spotlights on the problem," Cate said.
Noller said future research will proceed in two main directions. One goal is to solve the structure of the ribosome at higher and higher resolutions, eventually producing an atomic model of the entire complex. The other is to determine the structure of the ribosome in different stages of protein synthesis and with different molecules bound to it.
According to Yusupov, x-ray crystallography of ribosomes in different functional complexes will provide many interesting results. Interactions with antibiotics, many of which work by binding to and disrupting bacterial ribosomes, are also a promising area of research, Yusupov said. "In the future we can identify antibiotic binding sites, and that may lead to new kinds of drugs," he said.